Past Issues
Development of Nanoparticles Containing Bromealin
Íngara São Paulo1, Sandra A de Assis1, Fernanda Vidigal Souza2, Elaine M. Cabral-Albuquerque3, Cassia Britto Detoni4,*
1Enzymology and Fermentation Technology Laboratory, Health Department, State University of Feira de Santana, Feira de Santana, Brazil
2EMBRAPA Centro Nacional de Pesquisa de Mandioca e Fruticultura Tropical, Brazil
3Departamento de Engenharia Química, Escola Politecnica, Federal University of Bahia, Brazil
4Instituto de Ciências Farmacêuticas – Centro Multidisciplinar de Macaé, Federal University of Rio de Janeiro. Av. Aluizio da Silva Gomes, 50 - Novo Cavaleiros, Macaé - RJ, Brasil
*Corresponding Author: Cassia Britto Detoni, Instituto de Ciências Farmacêuticas – Centro Multidisciplinar de Macaé, Federal University of Rio de Janeiro. Av. Aluizio da Silva Gomes, 50 - Novo Cavaleiros, Macaé - RJ, Brasil, CEP 27930-560; Email: [email protected]
Received Date: January 23, 2024
Publication Date: March 20, 2024
Citation: Paulo IS, et al. (2024). Development of Nanoparticles Containing Bromealin. Dermis. 4(1):7.
Copyright: Paulo IS, et al. © (2024).
ABSTRACT
Bromelain is a protein complex containing proteases and other non-proteolytic constituents, with several therapeutic applications, such as: debriding, antineoplastic, anticoagulant and anti-inflammatory. The present work aimed to produce Eudragit® S100 nanoparticles (NP) containing bromelain extracted from a hybrid pineapple, Ananas comosus var. Erectifolius x Ananas comosus var. Bracteatus, characterize the nanocarrier and study optimal enzyme pH and temperature. NP were prepared by nanoprecipitation, using 2 polymer concentrations (1 and 2 mg/mL). Nanoparticles with adequate size (D[4,3] = 386 nm) and size distribution (Span= 0.6) were obtained when a lower polymer concentration was used. After adsorption, an increase in particle size (486 nm) and change the zeta potential (from -19 mV to -5.5 mV) was verified. Total protein quantification after immobilization showed a small loss. The association efficiency (AE%) of total protein was 45.24%, and the catalytic activity was maintained after the adsorption. A 3 Level Factorial Design was used to study the optimal pH (7.15) and temperature (52.5 °C) for free enzyme, and optimal pH (6.77) and temperature (52.5 °C) for adsorbed bromelain. This work revealed the viability of bromelain immobilization by adsorption to Eudragit S100 nanoparticles, which had a protective effect on the enzymatic activity during storage.
INTRODUCTION
Bromelain is a complex of proteases found in pineapples (Ananas comosus), which belong to the class of hydrolases (proteases that break the peptide bond of proteins and peptides) [1]. It is a mixture of different compounds, such as, proteases, phosphatases, peroxidases, cellulases, protease inhibitors, and others, besides carbohydrates and other not identified components [2]. Different bromelain applications are described in the medical field. It has aroused particular interest in plastic surgery due to its anti-edematous, anti-inflammatory and anticoagulant activities. In addition bromelain has been applied, to reduce pain, assist in wound healing, debridement of burns, ischemia and reperfusion, and as an adjuvant in antibiotic therapy [2,3]. The medicinal properties of pineapple plant are recognized traditionally in South America, China and Southeast Asia. Today, some of these properties are attributed to presence of bromelain in this plant. Among its various medicinal properties, bromelains anti-cancer activity has gained attention among researchers. Studies have shown that bromelain induces tumor cell apoptosis [4]. Its anti-inflammatory effect has been demonstrated in different animal models of inflammation and inflammatory human diseases, including arthritis and inflammatory bowel disease [5].
The topical use of bromelain rapidly hydrolysis eschar from burns with minimal injury to normal tissue, and it was demonstrated that the enzymatic debridement using the enzyme topically, accelerates the recovery of blood perfusion in injured tissues, and controls TNF-α expression, a cytokine involved in the inflammatory response [6]. It is effective in debridment of uncomplicated firearm wounds and is used as an adjuvant to conventional surgical debridement [7]. The nanoencapsulation of bromelain, together with vitamin E, has showed better activity to heal burnt skin on the animal model [8].
Polymeric nanoparticles emerged as a promising alternative as drug carrier. These systems have high surface area and are considered vectors for the delivery of substances, allowing a controlled release and often homogeneous drug release, thus increasing the therapeutic response at the site of action for a prolonged period [9]. These carrier systems are associated to the drug molecule by encapsulation or/and adsorption to the polymeric structure. Nanoencapsulation of bromelain has the main purses of stabilizing the proteolytic complex [10].
The use of poly(lactic-co-glycolic) acid nanoparticles to load bromelain has been previously reported [4]. In this work bromelain was associated by encapsulation [4], which is particularly difficult considering that these particles are more easily linked to lipophilic molecules [11]. Nanoparticles can absorb proteins on their surface by interactions composed of different contributions (hydrophobic, electrostatic, and H- bonding) [12]. According to Arumugam and Ponnusamy (2013) [13] the bromelain surface charge of is positive. The electrostatic interaction of this enzyme with a negative nanoparticle surface was exploited in this paper to produce nanoparticles adsorbed with bromelain.
Eudragit S100® is a pH-sensitive anionic copolymer composed of methacrylic acid and methylmethacrylate having unique dissolution behavior above pH 7. Many authors reported the use Eudragit nanostructures to enhance the drug delivery [14-19].
The nanoparticles made of Eudragit® S100 can be a potential carrier for not only topical delivery but also systemic delivery of therapeutically active compounds including microbicides, peptides and proteins [17].
The aim of this work was to produce and characterize Eudragit® S100 nanoparticles associated with bromelain from the pulp of a new variety of pineapple obtained by hybridization between Ananas comosus var. Erectifolius and Ananas comosus var bracteatus.
MATERIALS AND METHODS
CHEMICALS AND PLANT MATERIALS
Pineapples were obtained from the Active Germplasm Bank of Brazilian Agricultural Research Corporation (EMBRAPA – Empresa Brasileira de Pesquisa Agropecuária), in Cruz das Almas, Brazil. It is a new variety obtained from the hybridization between Ananas comosus var. erectifolius and Ananas comosus var. bracteatus. The Coomassie Brilliant Blue G-250 was obtained from Merck (Frankfuter, Germany). Eudragit S100 was kindly donated by Evonik® Brasil, SP-Brazil. Polyssorbate 80 was obtained from Vetec Quimica Fina Ltda, RJ-Brazil. All the buffers and reagents used were of analytical grade.
PREPARATION OF ENZYMATIC EXTRACT
Pineapples were washed and air dried. Peel and crown were removed. The pulp was sliced into small pieces before it was homogenized in a domestic blender with cold phosphate buffer (0.2 M pH 6.0) at 1:1 (w:v) ratio. The resulting blend was filtered in a cheese cloth and then centrifuged at 10,000×rpm at 4 °C for 20 min. The obtained supernatant was frozen until the time of analysis and was called bromelain extract [20].
PREPARATION OF NANOPARTICLES
Polymeric nanoparticles (NP) were prepared by nanoprecipitation of a preformed polymer, as proposed by Fessi et al. in 1989 [21]. An anionic polymethacrylate was used (Eudragit® S100). Briefly, the polymer was solubilized in acetone and added, in a laminar flow, into an aqueous phase containing polyoxyethylenesorbitan monooleate (Tween 80) under magnetic stirring. Organic solvent was removed and the volume of water was adjusted by rota-evaporation. Bromelain extract was added dropwise in the suspension of NP under moderate magnetic agitation in order to establish an electrostatic interaction. The bromelain content and enzymatic activity were quantified after adsorption.
CHARACTERIZATION OF NANOPARTICLES
Formulated NPs were characterized physical-chemically regarding: size, morphology, protein content, association efficiency, zeta potential, pH and stability. For these particles, the zeta potential (Nano-ZS Zetasizer®, Malvern) is an essential parameter because the change in zeta potential indicates whether the association was successful. Mean particle size and distribution were measured by laser diffractometry (Mastersizer®/Malvern). Particle size reported was the average of three samples.
Association efficiency (AE%) was evaluated by determining the protein content (C), which was measured using Bradford´s method (1976) [22] as described below. In order to determine AE% the formulation was submitted to the ultra-filtration centrifugation
technique. The nanostructures were retained in the filter (100 kDa) and the unabsorbed protein content was quantified in the ultra-filtrate (UF). Association rate was indirectly calculated by applying the equation [23]:
AE% = (C-UF)/C x 100
The pH was measured to ensure that it is in the bromelain activity range.
Determination of pH was performed with a potentiometer (B474 Micronal). Stability was investigated in free bromelain and bromelain adsorbed NP. The samples were stored in refrigerator at 6 °C. Samples stability was measured by enzymatic activity. All analyzes were performed in triplicate and statistical difference was evaluated by Student´s t-test.
Scanning electronic microscopy images were obtained on a JEOL JSM 6060 at 15 kV. Samples were coated with a thin layer of gold before visualization. The experiment was carried out at the Centro de Microscopia Eletrônica of the Federal University of Rio Grande do Sul, Porto Alegre, Brazil.
PROTEIN CONTENT AND ENZYMATIC ACTIVITY
Samples were diluted four times in ultrapure water and centrifuged in ultrafilters (100 kDa). In order to guaranty that bromelain did not interact with the filter material free bromelain was also filtrated and the protein content in the ultrafiltrate measured.
Protein content was spectrophotometrically measured according to Bradford (1976) [22], bovine serum albumin was used as standard.
Enzymatic activity was experimentally determined on the formulation, the ultrafliltrate, as well as on the retained fraction of the ultrafilter, considering that the adsorption of the protein can interfere on the protein activity due to steric impendent.
Enzyme activity was estimated as follows, according to modifications in the method described by Murachi (1976) [24]: 1% casein (w/v) in 50 mM phosphate buffer (pH 6.0) was used as substrate. Sample aliquots of 50 µL were added to tubes containing 500 µL casein buffered solution. The mixture was kept in water bath at 37 °C for 20 minutes. Subsequently, a 40 µL aliquot was removed from the reaction and added to 2 mL Bradford reagent. Absorbance was determined at 595 nm using UV/visible spectrophotometer (Varian Cary). The same mixture without reaction was used as control. One enzyme unit (U) was defined as the bromelain amount necessary to consume 1 µg of casein in 1 min (expressed as U/mL) at 37 °C. All measurements were performed in triplicate.
CHARACTERIZATION OF BROMELAIN OPTIMUM pH AND TEMPERATURE
A surface response three level factorial design was used to determinate optimum pH and temperature of free bromelain and NP adsorbed with bromelain. Each variable was studied at 3 levels, pH ranged from 5 to 7 and temperature ranged from 30 to 90°C.
Three central point (c) repetitions were performed to estimate the possible pure error in a total of eleven experiments (Table 1). Proteolytic activity was measured at each point.
ANOVA - variance analysis and response surface graphs were plotted using Statistica software, version 7.0.
Table 1: Three Level Factorial Design used to characterize bromelain extract obtained from a hybrid pineapple (Ananas comosus var. Erectifolius x Ananas comosus var. Bracteatus).
|
Assay |
pH |
Temperature (°C) |
|
1 |
5 |
90 |
|
2 |
6 |
90 |
|
3 |
7 |
90 |
|
4 |
5 |
60 |
|
5 |
6 |
60 |
|
6 |
6 |
60 |
|
7 |
6 |
60 |
|
8 |
7 |
60 |
|
9 |
5 |
30 |
|
10 |
6 |
30 |
|
11 |
7 |
30 |
RESULTS AND DISCUSSION
Bromelain is found in different tissues of a pineapple plant [25]. Its empirical use, with medicinal purpose, is known since 1493, when the pineapple extract was used by the Central and South American native population to treat different diseases. Today, its therapeutic actions are attributed to bromelain [26]. In this work, bromelain was extracted of a new variety of pineapple obtained by hybridization between Ananas comosus var. erectifolius and Ananas comosus var. bracteatus.
The obtained bromelain extract was immobilized on Eudragit® S100 NPs by adsorption. The polymer/extract interaction can be attributed to a physical attraction, electrostatic adsorption. Inert polymers and inorganic materials are usually used as support for the immobilization of proteins. Immobilization is a confinement of the protein to a phase (matrix/support) different from the substrates and products. This strategy prevents the aggregation, proteolysis and interaction of proteins with hydrophobic interfaces [27]. The protein-NP conjugate is a reversible immobilization and the enzyme can be detached from the support under gentle conditions [28].
NANOPARTICLESCHARACTERIZATION
Size, Zeta Potential and Morphology of NPs
Synthetic organic polymers have the great variability on physical and chemical characteristics. The main synthetic polymers are polystyrene, polyacrylate, polyvinyls, polyamide, polypropylene and copolymers are polyaldehyde and polypeptide structures [29]. In this work, nanoparticles were produced by nanoprecipitation of Eudragit® S100, an anionic polymer.
The prepared nanoparticles were nanometric, having mean diameter of 887 nm for the higher concentration of Eudragit® S 100 and 386 nm for the lower. Particle size is an important factor to determinate the biodistribution of circulating NPs and ensures the therapeutic efficacy [10]. Using a different technique of NPs production, a modified quasi-emulsion solvent diffusion method, Yoo, Giri and Lee (2011) [17] obtained Eudragit® S100 NPs with size 211.5 nm. While Nepolean et al. (2012) [18] used a cationic polymer (Eudragit® RLPO) to produce NPs and obtained particles with size from 496 to 984 nm, increasing according the increment in polymer concentration.
These results are similar with our results, smaller NPs are obtained when using a lower polymer concentration. Other report used papain, a cysteine protease, like bromelain, loaded in submicron particles of Eudragit® S100 and L100. The particle size varied 242 from 665.6 nm to 2785.6 [30].
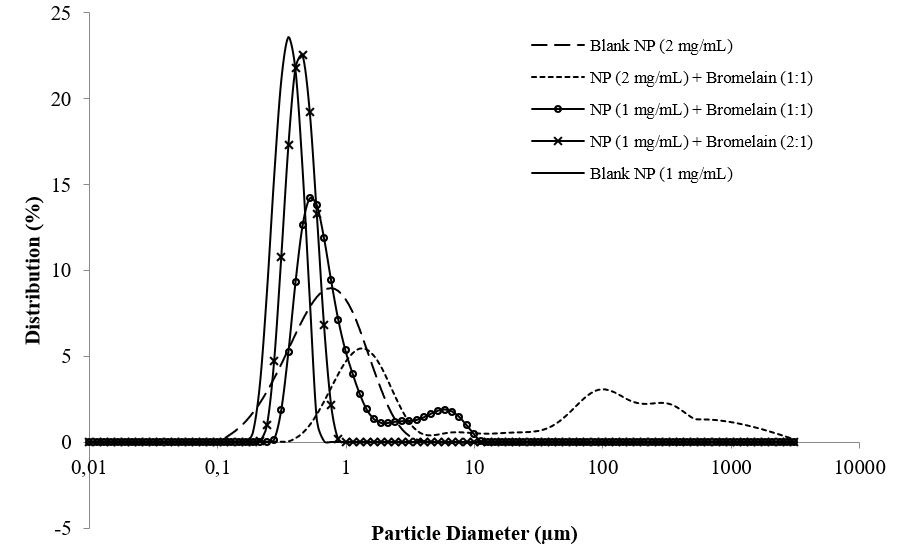
A monomodal size distribution with Span of 0.665 was observed for the formulation with 1 mg/mL of Eudragit® S100 with 2:1 ratio NP:bromelain but when a higher proportion of bromelain was added a second larger population was also formed (Fig. 1). This indicates an excess of bromelain when the ratio is 1:1 NP:bromelain.
When bromelain was added to the NPs with higher polymer concentration, aggregate were formed and the resulting suspension was multimodal (Fig. 1). The different behavior of the protein/NP interaction can be attributed to the difference in particle size. According to Aubin-Tam and Hamad-Schifferli (2008) NP size can affect the protein structure and activity [31]. Possibly, the observed result indicates that when interacting with particles of higher mean diameter, bromelain alters its structure and precipitates in larger agglomerates, but the interaction with particles of mean diameter 386 nm resulted in the successful adsorption of the protein.
Figure 1: Size distribution of blank Eudragit® S100 nanoparticles and NPs associated with bromelain extracted from pulp of a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
Table 2. Nanometric formulations of free Eudragit® S100 nanoparticles and NPs associated with bromelain extracted from pulp of a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
|
Polymer concentration (mg/mL) |
Ratio nanoparticle:bromelain |
D[4,3] (μm) |
Span |
Zeta Potencial (mV) |
pH |
|
1 |
(1:0) |
0.387±0.008 |
0.621±0.04 |
-19.0±0.85 |
5.6 |
|
1 |
(2:1) |
0.496±0.009 |
0.662±0.02 |
-5.5±0.39 |
5.6 |
The zeta potentials of the blank NPs were initially highly negative (-19.0 mV), after the addition of bromelain the negative charge was lower (-5.5 mV). This shows that the particle surface charge suffered changed once bromelain was added, indicating an electrostatic interaction. The results indicate that the bromelain was adsorbed on NP surface once the size of the NP increased after the bromelains addition besides the change on zeta potential (Table 2).
Morphology of nanoparticles associated to bromelain (2:1) was evaluated by scanning eletron microscopy (Fig. 2). Nanoparticles presented a spherical homogenous format.
Figure 2: Scanning electron microscopy of Eudragit S100 nanoparticles associated with bromelain extract from a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
Protein content was measured in order to determine the AE% and the maintenance of enzymatic activity after adsorption was also evaluated (Table 3).
Table 3. Protein content and proteolytic activity on formulations containing Eudragit® S100 NPs associated with bromelain extracted from pulp of a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
|
Sample |
Protein Content (µg/mL) |
Proteolytic activity (U/mL) |
|
Bromelain |
496.0 ± 6.7 |
44.1 ± 2.7 |
|
NP + bromelain |
425.04 ± 8.94 |
47.91 ± 0.82 |
|
Ultrafiltrate |
232.75 ± 10.00 |
41.71 ± 3.38 |
|
Retained fraction |
- |
32.71 ± 1.16* |
*statically different than bromelain before adsorption (p < 0.05; Student´s t-test). The AE% was 45.24% with maintenance of enzymatic activity. Different efficiency has been found by other authors [34] when producing encapsuled papain on submicron Eudragit® S100 e L100 particles obtaining an encapsulation efficiency of 81.17% and 82.35%, respectively. BHATNAGAR et al. (2014 and 2015) [4,10] reported Eudragit® coated bromelain loaded poly(lactic-co-glycolic) acid NPs with encapsulation efficiency of 48.8% and 52%, respectively. Adsorption usually renders lower AE% than encapsulation, even so the AE% for these particles is close to the ones found by Bhatnagar and co-workers for the same protein.
Some therapeutic activities of bromelain are based on its effects on the production of cytokines and inflammatory mediators, these effects require that bromelain be proteolytically active [5]. One of the advantages of using physical interaction between an enzyme and an polyelectrolyte, such as Eudragit® S100, is that the native structure and function of the protein is not markedly lowered [31]. The proteolytic activity of the retained fraction after the ultra-filtration was lower (p< 0.05) than the NP formulation and the bromelain pulp extract. A reduction of enzyme activity was expected, considering an AE% of 45.24%. The ultrafiltrate presented a proteolytic activity equal to the bromelain pulp extract and NP formulation. The pulp extract of bromelain contains several proteinase inhibitors [32], there possibly occurred the immobilization of proteases inhibitors in the formulation or filter membrane, rendering an ultrafiltrate with low or no proteases inhibitors.
The enzyme stability was determined by quantifying its activity on bromelain and the formulation (NPs + bromelain) for 0, 1, 3 and 5 days. After 1 and 3 days at 6 °C, the free bromelain retained 38.6% ± 2.2% and 17.2% ± 0.3% of its proteolytic
activity loosing completely its activity after 5 days. The bromelain associated to NPs retained 55.5% ± 2.8% and 27.2% ± 1.8%, respectively, after 1 and 3 days, with maintenance of 5.3% ± 0.9% of its activity at the last day. Similar result was reported by PARODI et al. (2014) [33] that investigated bromelain's proteolytic activity by storing it 24 h at 37°C. The extract displayed significant autodigestive activity. The same autodigestive effect was observed on our bromelain but less loss was observed on associated bromelain to NPs. This immobilization had a protective effect on the biocatalyst.
THE EFFECT OF TEMPERATURE AND pH ON THE PROTEOLYTIC ACTIVITY OF BROMELAIN
Temperature and pH are very important characteristics in enzyme kinetics studies. These features affect directly the enzymatic behavior and its stability. The studied temperature ranged from 30 to 90 °C and pH from 5 to 7 as showed on Table 1.
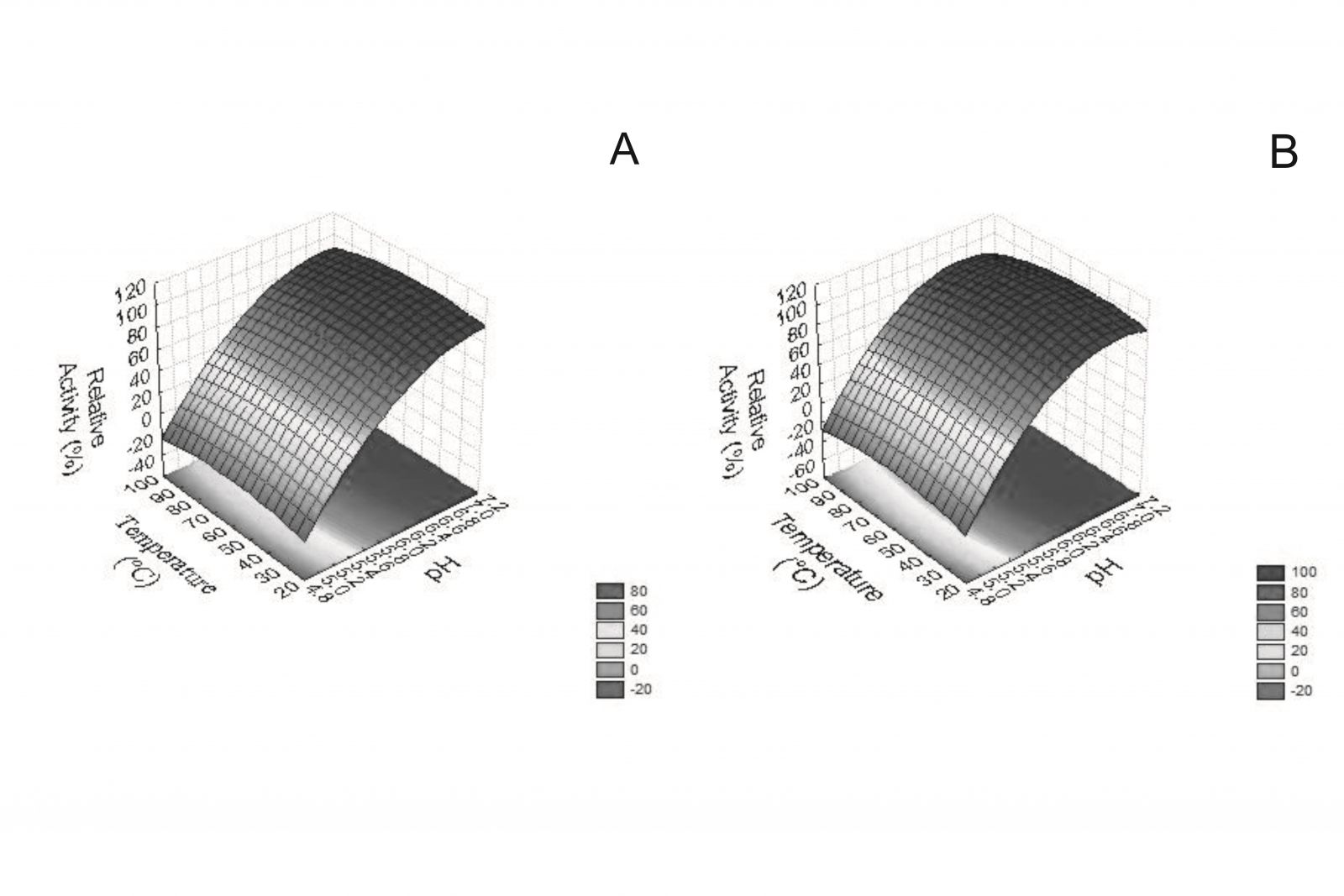
Results were obtained in U/mL and were plotted in relative activity to better display the bromelain kinetic reaction features under temperature variations. As a consequence of enzyme immobilization, some properties such as catalytic activity or thermal stability become altered [28]. To ensure the interference of immobilization on enzymatic kinetics, the optimum pH and temperature were quantified the formulation containing bromelain associated to Eudragit® S100 NPs. A Three Level Factorial Design with two variables was used for this characterization. Response surface plots are shown in Fig. 3A and 3B.
Figure 3: Kinetic characterization of free bromelain (A) and Eudragit® S100 NPs Containing bromelain (B) from pulp of a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
Results show that bromelain has maximum activity at 52.5 °C and pH 7.15 while the bromelain NP have its maximum catalytic capacity at the same temperature and pH 6.77. This little difference on pH values is not significant. Immobilization of biocatalysts can protect them from changes in their 3-D structure face to experimental variations. The experimental matrix applied in the current study described the well- adjusted behavior of bromelain activity against the variables analyzed. Variance analysis, presented in Table 3, show that the results are statistically significant and that there was no lack-of-fit. The model’s efficiency was checked by determining the coefficient R2 0.998 and 0.9976 for free and associated bromelain, respectively. This indicates a good match between the response predicted by the model and the experimentally obtained enzymatic activity. A test based on Fisher distribution (F-test) indicates that there is good agreement between the models predicted response and the experimental values studied for each variable. There is no lack-of-fit for kinetic characterization of free (F = 45.67 < 99.17) and adsorbed bromelain (F = 8.621 < 19.16)
Table 4: Variance analysis of kinetic characterization of free bromelain and Eudragit® S100 NPs containing bromelain from pulp of a hybrid pineapple Ananas comosus var. erectifolius x Ananas comosus var. bracteatus.
|
ANOVA |
||||||||||
|
Variation source |
Free bromelain |
NPs formulation with bromelain |
||||||||
|
SS |
DF |
MS |
Fcal |
Ftab |
SS |
DF |
MS |
Fcal |
Ftab |
|
|
Regression |
15059.4758 |
5 |
3011.895 |
597.6806 |
10.96702 |
16624.8710 |
5 |
3324.974 |
414.9716 |
5.05 |
|
Residual |
25.1965 |
5 |
5.03931 |
|
|
40.0627 |
5 |
8.01253 |
|
|
|
Lack-of-fit |
24.83 |
3 |
8.28 |
45.67 |
99.17 |
37.19 |
3 |
12.40 |
8.621 |
19.16 |
|
Pure error |
0.36 |
2 |
0.18 |
|
|
2.88 |
2 |
1.44 |
|
|
|
Total SS |
15084.67 |
10 |
|
|
|
16664.93 |
10 |
|
|
|
SS – sum of squares; DF – degree of freedom; MS – mean square; Fcal – calculated F value; Ftab –
tabulated F value.
Other authors reported higher bromelain activity at 59 °C [34], 40 °C [35] 30 °C with rapid activity loss above 60 °C [36] or 50 to 70 °C, with total loss of its proteolytic activity when exposed to 90 °C [37]. It is clear that the enzymatic kinetic features can vary depending on the source of the biomolecule. The pH variation is another essential parameter in enzyme feature studies. Changes on pH cause different ionization pattern on the biomolecule. These variations can enhance its affinity with its substrate or can cause its denaturation. Studies reported optimum pH of 7.7 [34], of 5.0 [35] and of 6.0 [36] and range between pH 6-7 [38] and 7 for a commercial bromelain [37].
CONCLUSION
It was possible to extract bromelain from the pulp of a hybrid pineapple (Ananas comosus var. Erectifolius x Ananas comosus var. Bracteatus). The bromelain extract was successfully adsorbed to Eudragit S100® nanoparticle as showen by an increase of particle size and decrease in zeta potential modulus. This association maintained the proteolytic activity of the enzymes, which is important for future therapeutic applications. Bromelain kinetic reaction features were also maintained when associated to the NP. Additionally, adsorption of bromelain to NP exerted a protective effect on enzymatic activity during storage.
REFERENCES
- Mynott TL, Ladhams A, Scarmato P, Engwerda CR. (1999). Bromelain, from pineapple stems, proteolytically blocks activation of extracellular regulated kinase-2 in T cells. J Immunol. 163:2568-2575.
- Hikisz P, Bernasinska-Slomczewska J. (2021). Beneficial properties of bromelain. Nutrients. 13(12):4313.
- Orsini RA. (2006) Safety & Efficacy Report: Bromelain. Plast Reconstr Surg. 118.
- Bhatnagar P, Pant AB, Shukla Y, Chaudhari B, Kumar P, Gupta KC. (2015). Bromelain nanoparticles protect against 7,12-dimethylbenz[a] anthracene induced skin carcinogenesis in mouse model. Eur J Pharm Biopharm. 91:35-46.
- Hale LP, Greer PK, Trinh CT, James CL. (2005). Proteinase activity and stability of natural bromelain preparations. Int Immunopharmacol. 5:783-793.
- Wu SY, Hu W, Zhang B, Liu S, Wang JM, Wang AM. (2012). Bromelain ameliorates the wound Microenvironment and improves the healing of firearm wounds. J Surg Res. 176:503-509.
- Hu W, Wang AM, WuSY, Zhang B, Liu S, Gou YB, Wang J. (2011). M. Debriding effect of bromelain on firewarm wounds in pigs. J Trauma. 71(4).
- Rachmawati H, Sulastri E, Iwo MI, Safitri D, Rahma A. (2016). Bromelain encapsulated in self assembly nanoemulsion exhibits better debridement effect in animal model of burned skin. J Nano Res. 40:158-166.
- Venturini CG, Jager E, Oliveira CP, Bernardi A, Battastini AMO, Guterres SS, et al. (2011). Formulation of lipid core nanocapsules. Colloids Surf A Physicochem Eng Asp, 75 (1-3):200-208.
- Ataide JA, Gérios EF, Mazzola PG, Souto EB. (2018). Bromelain-loaded nanoparticles: A comprehensive review of the state of the art. Adv Colloid Interface Sci. 254:48-55.
- Schaffazick S, Guterres S, Freitas L, Pohlmann A. (2003). Caracterização e estabilidade físico-química de sistemas poliméricos nanoparticulados para a administração de fármacos. Quím Nova. 26(5):726-737.
- Monopoli MP, Walczyk D, Campbell A, Elia G, Lynch I, Bombelli FB, Dawson KA. (2011). Physical-chemical aspects of protein corona: relevance to in vitro and in vivo biological impacts of nanoparticles. J Am Chem Soc. 133:2525–2534.
- Arumugam A, Ponnusami V. (2013). Pineapple fruit bromelain recovery using recyclable functionalized ordered mesoporous silica synthesized from sugarcane leaf ash. Braz J Chem Eng, 30(3):477-486.
- Leroux JC, Cozens RM, Roesel JL, Galli B, Doelker E, Gurny R. (1996). pH-sensitive nanoparticles: an effective means to improve the oral delivery of HIV-1 protease inhibitors in dogs. Pharm Res. 13(3):485-487.
- Jain D, Panda AK, Majumdar DK. (2005). Eudragit S100 entrapped insulin microspheres for oral delivery. AAPS Pharm Sci Tech. 6(1):E100-E107.
- Pereira R, Julianto T, Yuen KH, Majeed ABA. (2006). Anionic Eudragit nanoparticles as carriers for oral administration of peptidomimetic drugs. Brisbane, Austrália, International Conference on Nanoscience and Nanotechnology, ICONN:298-301.
- Yoo JW, Giri N, Lee CH. (2011). pH-sensitive Eudragit nanoparticles for mucosal drug delivery. Int J Pharm, 403:262-267.
- Nepolean R, Narayanan N, Subramaniyan N, Venkateswaran K, Vinoth J. (2002). Preparation and characterization of nisoldipine nanoparticles by nanoprecipitation method. J Pharm Sci & Res. 4(11):1989- 4301994.
- Subramanian L, Ganesan V, Venkateshan N, Solairaj P. (2012) Design, formulation and evaluation of ofloxacin nanoparticles. Int J Bio Pharm Res. 3(5):659-662.
- São Paulo Barretto Miranda ÍK, Fontes Suzart Miranda A, Souza FV, Vannier-Santos MA, Pirovani CP, Pepe IM, et al. (2016). The biochemical characterization, stabilization studies and the antiproliferative effect of bromelain against B16F10 murine melanoma cells. Int J Food Sci Nutr, p. 1-13.
- Fessi H, Puisieux F, Devissaguet JP, Ammoury N, Benita S. (1989). Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int J Pharm. 55(1):R1-R4.
- Bradford MM. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 72:248-254.
- Santos Maia C, Mehnert W, Schaller M, Korting HC. Gysler A, Haberland A, et al. (2000). Drug targeting by solid lipid nanoparticles for dermal use. Int J Pharm. 196:165-167.
- Murachi T. (1976). Bromelain Enzymes. In: Lorand L. Methods in Enzymology. New York: Academic Press. XLV:475-485.
- Ketnawa S, Chaiwut P, Rawdkuen S. (2012). Pineapple wastes: a potential source for bromelain extraction. Food Bioprod Process, 90:385-391.
- Taussig S, Batkin S. (1998). Bromelain, the enzyme complex of pineapple (Ananas comosus) and its clinical application: an update. J Ethnopharmacol. 22:191- 454
- Datta S, Christena LR, Rajaram YRS. (2013). Enzyme immobilization: an overview on techniques and support materials. Biotech, 3:1-9.
- Brena BM, Batista-Viera F. (2006). Immobilization of enzymes. In: Immobilization of enzymes and cells. 2ed. New Jersey: Humana Press Inc.
- Tischer W, Wedekind F. (1999). Immobilized enzymes: methods and applications. Top Curr Chem. 200:95-126.
- Sharma M, Sharma V, Panda AK, Majumdar DK. (2011). Development of enteric submicron particle formulation of papain for oral delivery. Int J Nanomedicine. 6:2097-2111.
- Aubin-Tam ME, Hamad-Schifferli K. (2008). Structure and function of nanoparticle–protein conjugates. Biomed Mater. 3(3):034001.
- Müller C, Leithner K, Hauptstein S, Hintzen F, Salvenmoser W, Bemkop-Schnürch A. (2013). Preparation and characterization of mucus-penetrating papain/poly(acrylic acid) nanoparticles for oral drug delivery applications. J Nanopart Res. 15:1-13.
- Maurer HR. (2001). Bromelain: biochemistry, pharmacology and medical use. Cell Mol Life Sci. 58:1234-1245.
- Parodi A, Haddix SG, Taghipour N, Scaria S, Taraballi F, Cevenini A, et al. (2014). Bromelain surface modification increases the diffusion of silica nanoparticles in the tumor extracellular matrix. ACS Nano. 8(10):9874-9883.
- Corzo CA, Waliszewski KN, Chanes JW. (2012). Pineapple fruit bromelain affinity to different protein substrates. Food Chem. 133:631-635.
- França-Santos A, Alves RS, Leite NS, Fernandes RPM. (2009). Estudos bioquímicos da enzima bromelina do Ananas comosus (abacaxi).Scientia Plena. 5(11).
- Khan RH, Rasheedi S, HAQ SK. (2003). Effect of pH, temperature and alcohols on the stability of glycosylated and deglycosylated stem bromelain. J Biosci. 28(6):709-714.
- Xue Y, Wu CY, Branford-White CJ, Ning X, Nie HL, Zhu LM. (2010). Chemical modification of stem bromelain with anhydride groups to enhance its stability and catalytic activity. J Mol Catal B: Enzym. 63:188-193.
 Abstract
Abstract  PDF
PDF